Part:BBa_K2239008
CBD-LDH
CBD-LDH (T7 promoter--lac operator--RBS--His-tag--LDH--CBD--T7 terminator)
This device codes for the LDH-CBD fusion protein.
Construct
The vector of LDH-CBD for its expression is pET-28x. It is formed by modifying the restriction enzyme sites EcoR I and Xba I of vector pET-28a.
The LDH sequence is retrieved from the GenBank. It is artificially synthesized and inserted into plasmid pUC57. The LDH gene is then cloned from the plasmid by PCR amplification. The restriction site BamH I is added to the upstream primer, and Hind III is added to the downstream primer.
The CBD sequence is retrieved from the GenBank. It is artificially synthesized and inserted into plasmid pUC57. The CBD gene is then cloned from the plasmid by PCR amplification, with the restriction site Hind III added to the upstream primer, and Xhol I added to the downstream primer.
Firstly, the LDH gene is inserted into the modified pET-28x at BamH I and Hind III, and CBD at Hind III and Xhol I, after proliferation in T3 vector. Then the whole gene fragment, T7 promoter--lac operator--RBS--His-tag--LDH--CBD--T7 terminator, is retrieved from this plasmid by PCR amplification, with prefix containing EcoR I, Not I and Xba I added on its upstream primer, and suffix containing Pst I, Not I and Spe I added on its downstream primer. The PCR product is then connected to pSB1C3 at EcoR I and Pst I.
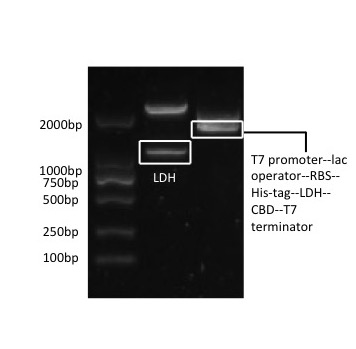
[Fig. 1. pSB1C3-CBD-LDH]
Usage and Biology
LDH(lactate dehydrogenase) catalyzes the conversion of lactate to pyruvic acid and back, as it converts NAD+ to NADH and back. It is used to catalyze the reduction of pyruvate into lactate, with the presence of NADH, which is oxidized into NAD+ at the same time.[1]
CBD (cellulose binding domain) is able to bind to cellulose. When connected to LDH, CBD is able to immobilize the enzyme LDH after expression, by binding to the gauze inside the solution on its cellulose.[2]
The function of cellulose binding domain
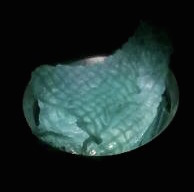
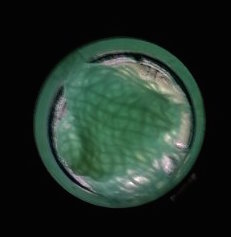
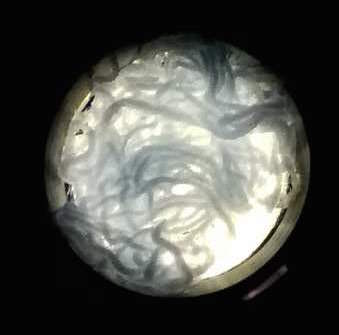
The function of CBD is tested by connecting CBD gene with GFP gene in pET28x. The GFP-CBD fusion protein is expressed and mixed with a gauze piece. The green fluorescent on the gauze is not significantly reduced after washing, proving that the CDB is well functioned. In comparison, no green fluorescent is left after washing the gauze mixed with GFP-ChBD (Chintin binding domain).
[Fig. 2. GFP-CBD on gauze before washing]
[Fig. 3. GFP-CBD on gauze after washing]
[Fig. 4. GFP-ChBD on gauze before washing]
[Fig. 5. GFP-ChBD on gauze after washing]
Expression and Immobilization[2]
The constructed pET28x-LDH plasmid is transformed into BL21(DE3) E.coli for expression. After that, when the OD 600 reached 0.6-0.8, 0.2mM IPTG is added in the liquid culture. The mixture is shaken at 20 ℃ overnight. The bacteria is collected by centrifugation at low temperature, 8000 rpm for 10 minutes, and the supernatant is discarded. The bacteria is then resuspended using 0.15M pH8.8 Tris-HCL, and is broken by ultrasonication.
The resulted bacteria solution is diluted to a certain concentration and mixed with gauze piece and the gauze piece is washed three times by ddH2O afterwards. As a result, the CBD protein binds to the cellulose on gauze, and the enzyme is successfully immobilized.
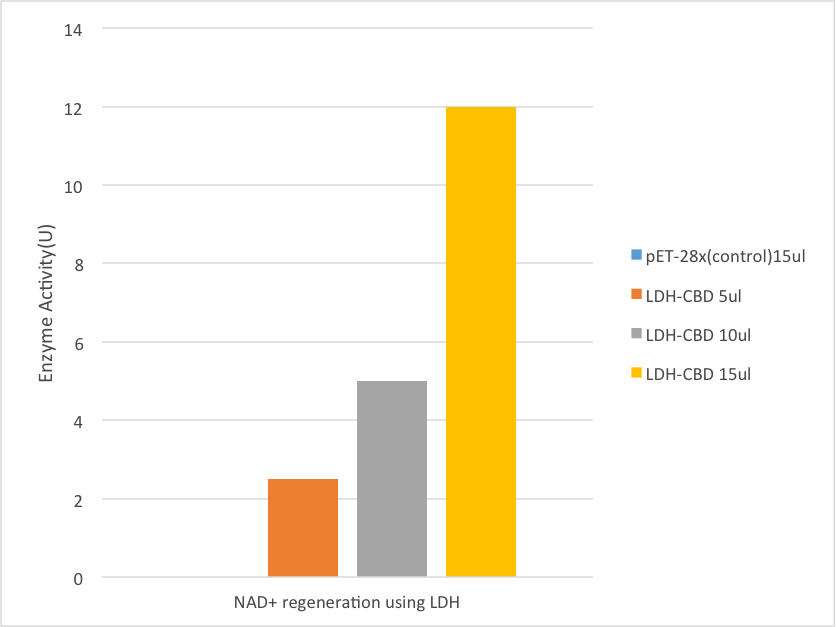
Enzyme Activity
The NAD+ regeneration
LDH is an NADH dependent enzyme from Lactobacillus delbrueckii subspecies bulgaricus. The 3mL reaction consists of 150mM phosphate buffer(pH 8.0), 25mM Lactose, 0.2mM NAD+. The reaction started when the solution is combined with LDH-CBD-enzyme-binding gauze that in different concentration, includes 5ul, 10ul, and 15ul liquid supernatant of ultrasonication bacteria solution. The control group was testify under the same solution and condition but using pET28x-CBD liquid supernatant of ultrasonication bacteria solution to bind with gauze in the concentration of 15ull. Before adding the gauze into the solution, the gauze was washed by ddH2O for 3 times in order to purify the enzyme.
The LDH works on pyruvate and take a pair of hydrogen(2H+and 2e-) from NADH to form lactate. The NAD+ was regenerate through this reaction. Since the NADH can be testified under 340nm of ultraviolet light, the reverse reaction of LDH is employed to testify its function. The enzyme activity was determined spectrophotometrically atv340 nm (ε = 6.22 mM-1 cm-1) and room temperature by measuring the synthesize of NADH. One unit of activity is defined as the amount of enzyme catalyzing the synthesize of 1 mmol of NADH per min under the assay conditions used.
Result
[Fig. 6. Result]
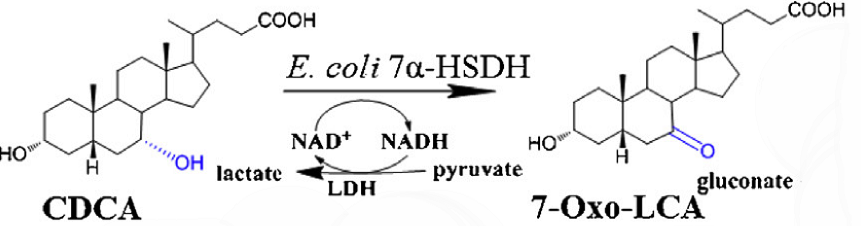
Oxidation of CDCA to 7-oxo-LCA using E. coli 7alpha-HSDH and NAD+ regeneration
[Fig. 7. Reaction process]
CDCA was converted in a 3mL solution containing 150 mM phosphate buffer(pH 8.0), 10 mM CDCA, 30 mM sodium pyruvate, 0.25mM NAD+, combined with 3U/ml LDH and1 1 U/ml E. coli DH5a 7a-HSDH-CBD at room temperature.
The bioconversion experiments were monitored via HPLC measurements. The sample was analyzed by UV detection at 210 nm, using a mobile phase of methanol–water mixture (final ratio 80:20,pH 3.5 with phosphoric acid) using chromatographic column C18.
Result
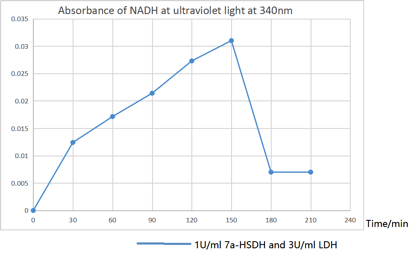
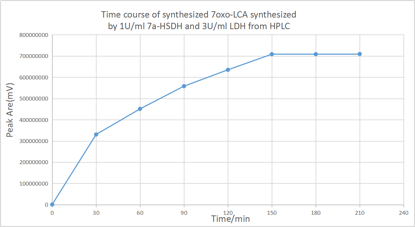
As shown in figure, the transformation was complete after 2.5h.
[Fig. 9. Absorbance of NADH]
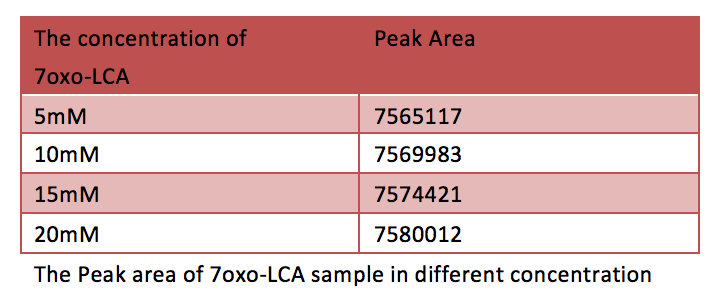
[Fig. 10. Peak area of 7oxo-LCA sample in different concentration]
[Fig. 11. Time course]
According to the HPLC after 150minute, there is no significant increase of 7oxo-LCA, as a result, most of the CDCA has been transformed into 7oxo-LCA. And according to the HPLC the final yielding rate is 94%.
According to the Absorbance of NADH that shown in figure, the absorbance is decreased significantly after 150 minute dual to the depleted CDCA that stop the conversion of CDCA to 7oxo-LCA and NADH synthesize. Because of the abundant amount of pyruvate in the solution, the LDH that works on pyruvate still regenerate the NAD+ by taking a pair of hydrogen from NADH until most of the NADH synthesized transformed into NAD+.
Reference
[1] Ming-Min Zheng, Ru-Feng Wang, Chun-Xiu Li, Jian-He Xu: Two-step enzymatic synthesis of ursodeoxycholic acid with a new 7β-hydroxysteroid dehydrogenase from Ruminococcus torques. Process Biochemistry, Elsevier, 2015.
[2] Etai Shpigel, Arie Goldlust, Gilat Efroni, Amos Avraham, Adi Eshel, Mara Dekel, Oded Shoseyov: Immobilization of Recombinant Heparinase I Fused to Cellulose-Binding Domain, 1999.
Sequence and Features
- 10COMPATIBLE WITH RFC[10]
- 12INCOMPATIBLE WITH RFC[12]Illegal NheI site found at 1266
- 21INCOMPATIBLE WITH RFC[21]Illegal BglII site found at 862
- 23COMPATIBLE WITH RFC[23]
- 25COMPATIBLE WITH RFC[25]
- 1000INCOMPATIBLE WITH RFC[1000]Illegal SapI site found at 1122
| None |